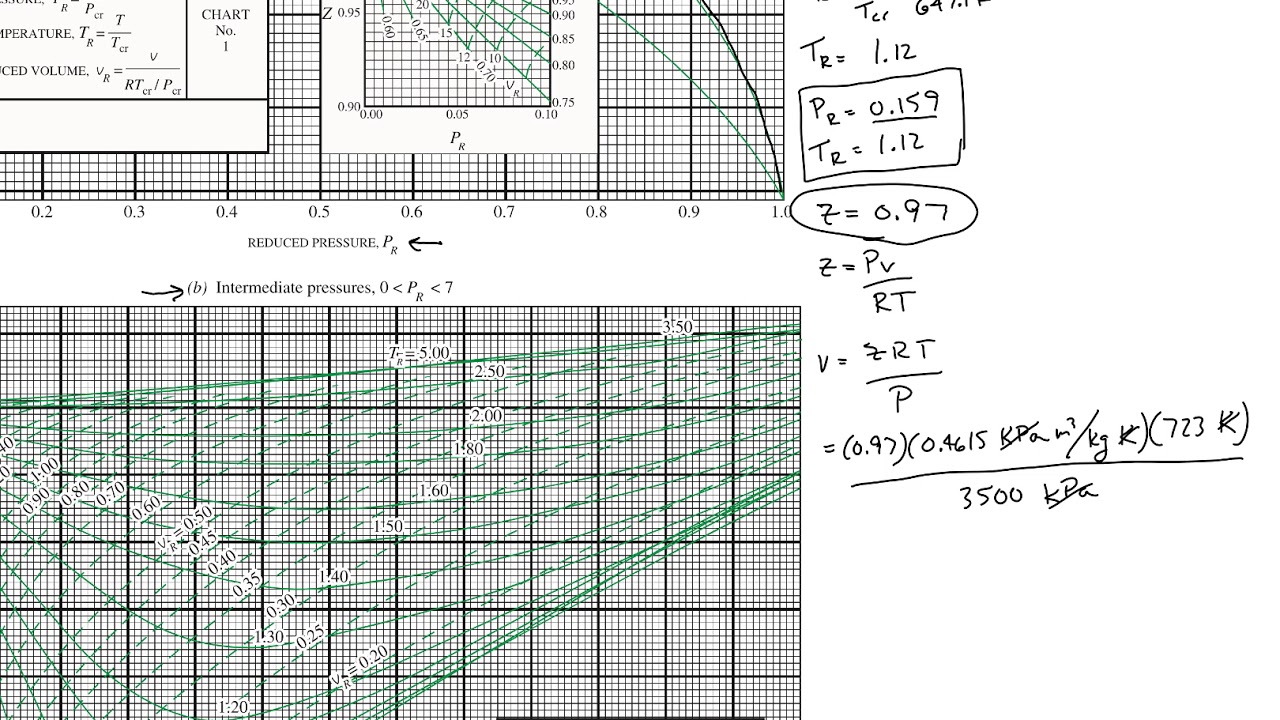
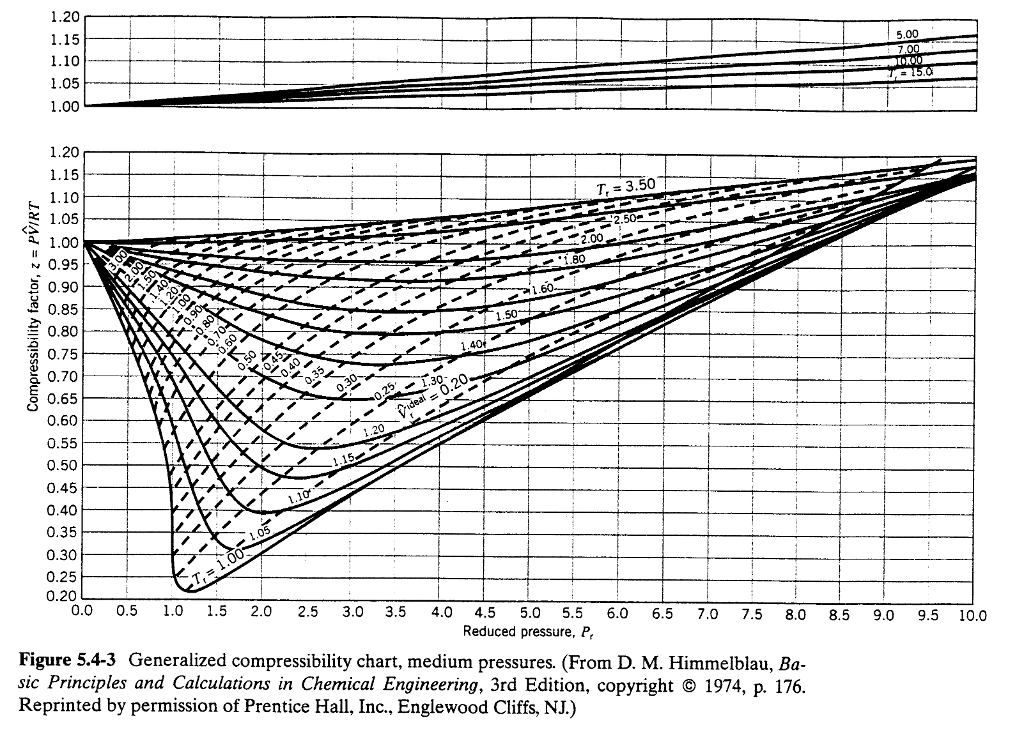
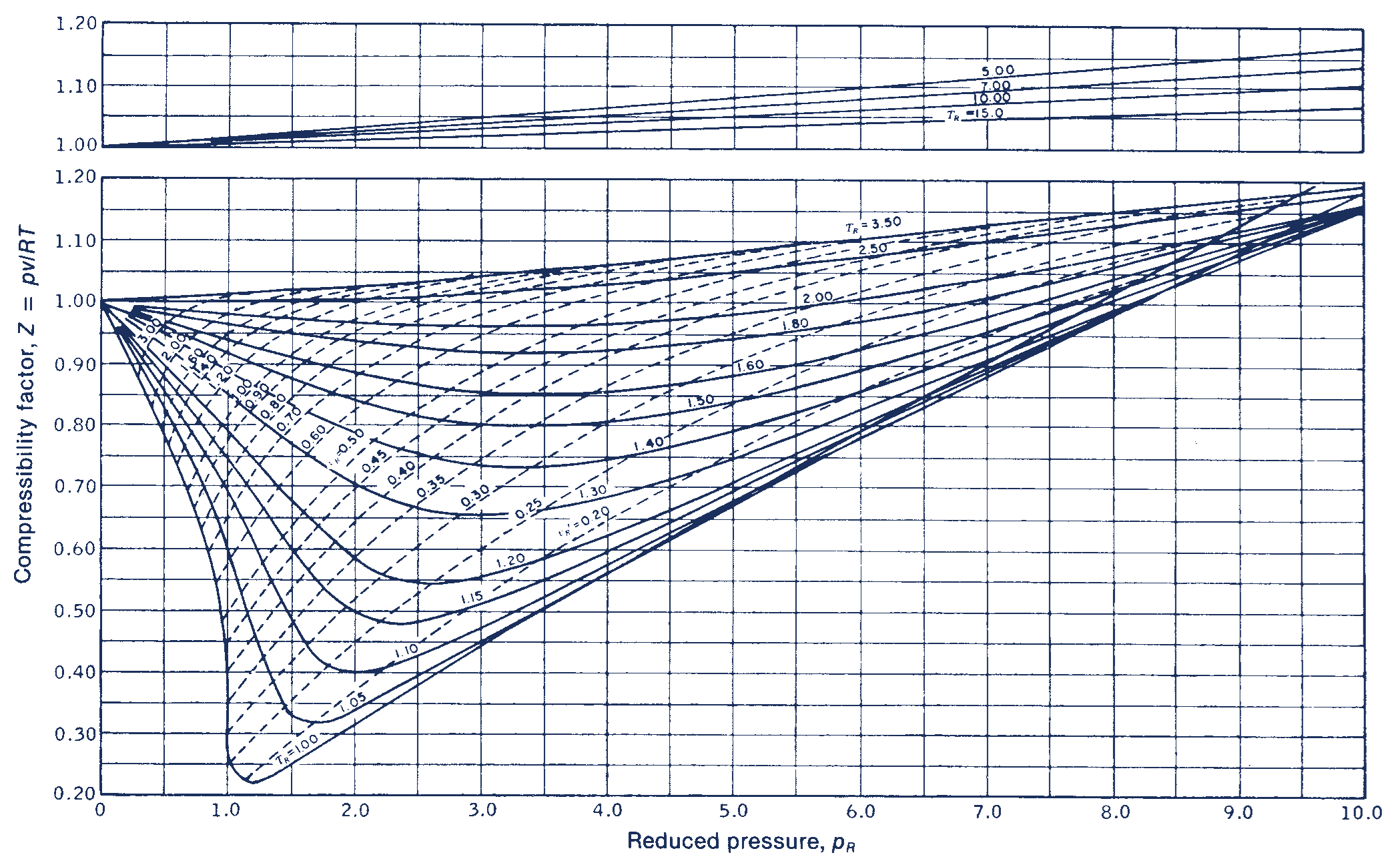
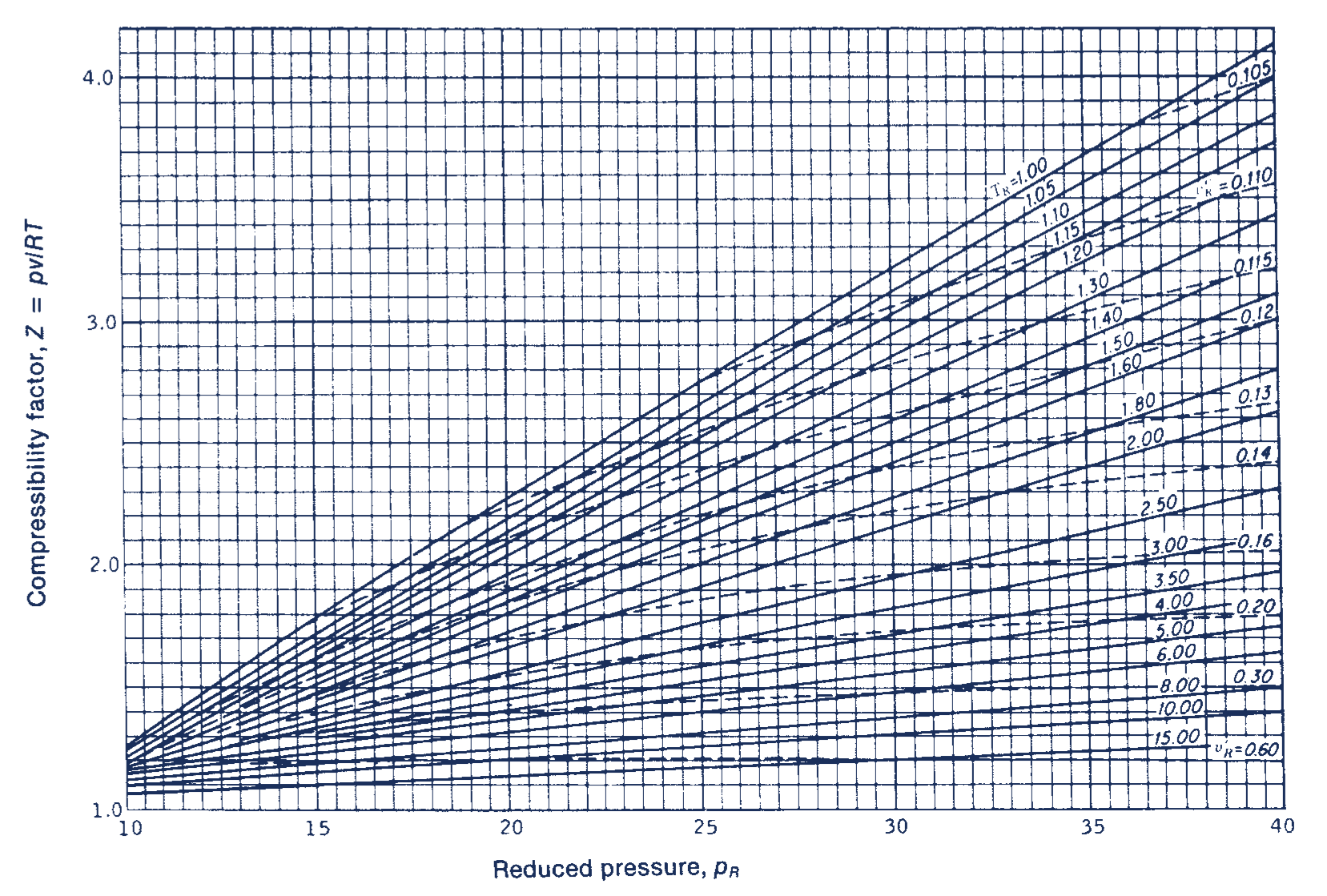
Generalized Compressibility Chart
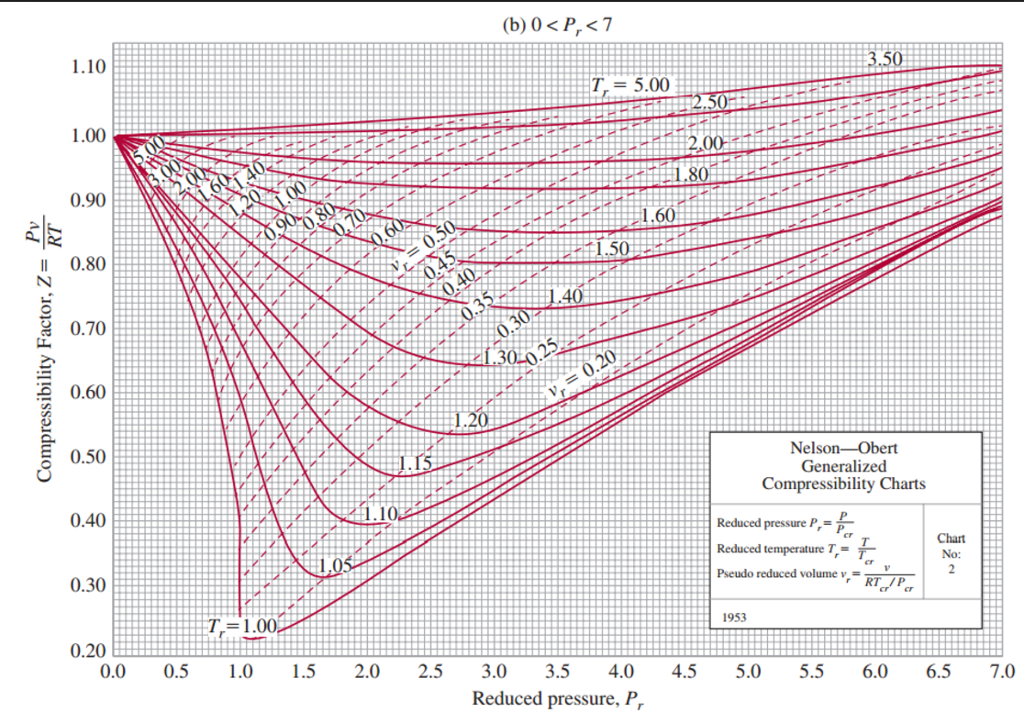
Generalized Compressibility Chart - Web the compressibility factor chart plots the compressibility factor , equal to , where is the volume per mole, versus the reduced pressure for several values of the reduced temperature. Web the compressibility factor is given by: This chart brings the following information: A test for whether a gas behaves ideally can be obtained by comparing the actual pressure and temperature to the critical pressure and temperature. 2.1 and 2.3) is modified for use for real gases by introducing the generalized compressibility factor, which is represented. If we only know the temperature and pressure, we can still calculate it using a compressibility chart. Values for p c and t c for various substances can be found in table c.12. Web properties of common gases. When p, pc, t, tc, v , and r are used in consistent units, z, pr, and tr are numerical values without units. Web 13.5.1 generalized compressibility chart. These have been extended [see, e.g.,. (b) the pressure in mpa at the final state. Vapor pressure curves for common pure gases. A practical guide to compressor technology, second edition, by heinz p. Reduced pressure is the ratio of the actual pressure. The reduced pressure and temperature are defined by and , respectively, where is the critical pressure and is the critical temperature. Compare the results of parts (a) and (b) with values obtained from the thermodynamic table or software11. 13.12 and 13.15 resulting in the following equations for real gases. Web the resulting z = z (p r, t r, v′ r) plot is now called the generalized compressibility chart and is shown in figures 11.5, 11.6, and 11.7. Then, a compressibility factor (z) can be used to quantify This chart brings the following information: Web figure 1 shows the essential features of a generalized compressibility factor chart. Vapor pressure curves for common pure gases. Web the compressibility factor is given by: Values for p c and t c for various substances can be found in table c.12. Web essentially it corrects for the deviation of a real gas from an ideal gas. At very low pressure (pr << 1), gases behave as an ideal gas regardless of temperature. Web the compressibility factor equation can be written as: Then, a compressibility factor (z) can be used to quantify The ideal gas equation (eq. Web the generalized compressibility chart can be viewed as a graphical representation of the gas behaviour over a wide range of pressures and temperatures. It is valid for many substances, especially those that have simple molecular structures. Z = p × v / n × r × t, where z is the compressibility factor, for pressure p, volume v, gas. Is the same for all gases. Z = p × v / n × r × t, where z is the compressibility factor, for pressure p, volume v, gas constant r, number of moles n, and temperature t. A practical guide to compressor technology, second edition, by heinz p. It is valid for many substances, especially those that have simple. Is the same for all gases. Web the compressibility factor equation can be written as: Web using the compressibility chart, determine (a) the specific volume of the water vapor in m3/kg at the initial state. For air at 200 k, 132 bar, tr = 200 k/133 k = 1.5, pr = 132 bar/37.7 bar =. 2.1 and 2.3) is modified. Web essentially it corrects for the deviation of a real gas from an ideal gas. The ideal gas equation (eqs. Web the resulting z = z (p r, t r, v′ r) plot is now called the generalized compressibility chart and is shown in figures 11.5, 11.6, and 11.7. The ideal gas equation (eq. 13.12 and 13.15 resulting in the. Web essentially it corrects for the deviation of a real gas from an ideal gas. At very low pressure (pr << 1), gases behave as an ideal gas regardless of temperature. Z = pv¯¯¯¯ rt z = p v ¯ r t. Web the compressibility factor chart plots the compressibility factor , equal to , where is the volume per. The ideal gas equation (eq. Then, a compressibility factor (z) can be used to quantify Reduced pressure is the ratio of the actual pressure. It is valid for many substances, especially those that have simple molecular structures. This chart brings the following information: Web the generalized compressibility factor chart shows how the value of {eq}z {/eq} fluctuates in regard to reduced pressure and temperature. Web generalized compressibility chart and the compressibility factor, z. Values for p c and t c for various substances can be found in table c.12. Is the same for all gases. At very low pressure (pr << 1), gases. Web the compressibility factor is given by: Milton beychok) we can rewrite the universal expression for compressibility \(z\) using reduced variables and plot measured values of \(z\) versus the reduced pressure, \(p_r\) (see figure 16.4.1 ). The reduced pressure and temperature are defined by and , respectively, where is the critical pressure and is the critical temperature. Bloch copyright ©. Example of a generalized compressibility factor graph (public domain; 2.1 and 2.3) is modified for use for real gases by introducing the generalized compressibility factor, which is represented. Reduced pressure is the ratio of the actual pressure. Web using the compressibility chart, determine (a) the specific volume of the water vapor in m3/kg at the initial state. Web the generalized compressibility factor chart shows how the value of {eq}z {/eq} fluctuates in regard to reduced pressure and temperature. At very low pressure (pr << 1), gases behave as an ideal gas regardless of temperature. Is the same for all gases. Values for p c and t c for various substances can be found in table c.12. Milton beychok) we can rewrite the universal expression for compressibility \(z\) using reduced variables and plot measured values of \(z\) versus the reduced pressure, \(p_r\) (see figure 16.4.1 ). On a generalized compressibility chart, the compressibility z z is plotted as a function f = f(pr,tr) f = f ( p r, t r) of the reduced pressure and temperature. At high temperatures (tr > 2), ideal gas behavior can be assumed with good accuracy. The ideal gas equation (eqs. Then, a compressibility factor (z) can be used to quantify Bloch copyright © 2006 john wiley & sons, inc. (b) the pressure in mpa at the final state. Compare the results of parts (a) and (b) with values obtained from the thermodynamic table or software11.Thermodynamics Generalized Compressibility Chart YouTube
Solved Use the generalised compressibility chart to estimate
Generalized compressibility charts Big Chemical Encyclopedia
Generalized Compressibility Chart
Generalized Compressibility Chart
Generalized Compressibility Chart
Generalized Compressibility Chart PDF Thermodynamics
Introduction to the Generalized Compressibility Chart Engineering
Generalized Compressibility Chart
Generalized Compressibility Chart Calculator A Visual Reference of
Z = P × V / N × R × T, Where Z Is The Compressibility Factor, For Pressure P, Volume V, Gas Constant R, Number Of Moles N, And Temperature T.
Web The Generalized Compressibility Chart Can Be Viewed As A Graphical Representation Of The Gas Behaviour Over A Wide Range Of Pressures And Temperatures.
Web 13.5.1 Generalized Compressibility Chart.
Web The Resulting Z = Z (P R, T R, V′ R) Plot Is Now Called The Generalized Compressibility Chart And Is Shown In Figures 11.5, 11.6, And 11.7.
Related Post: