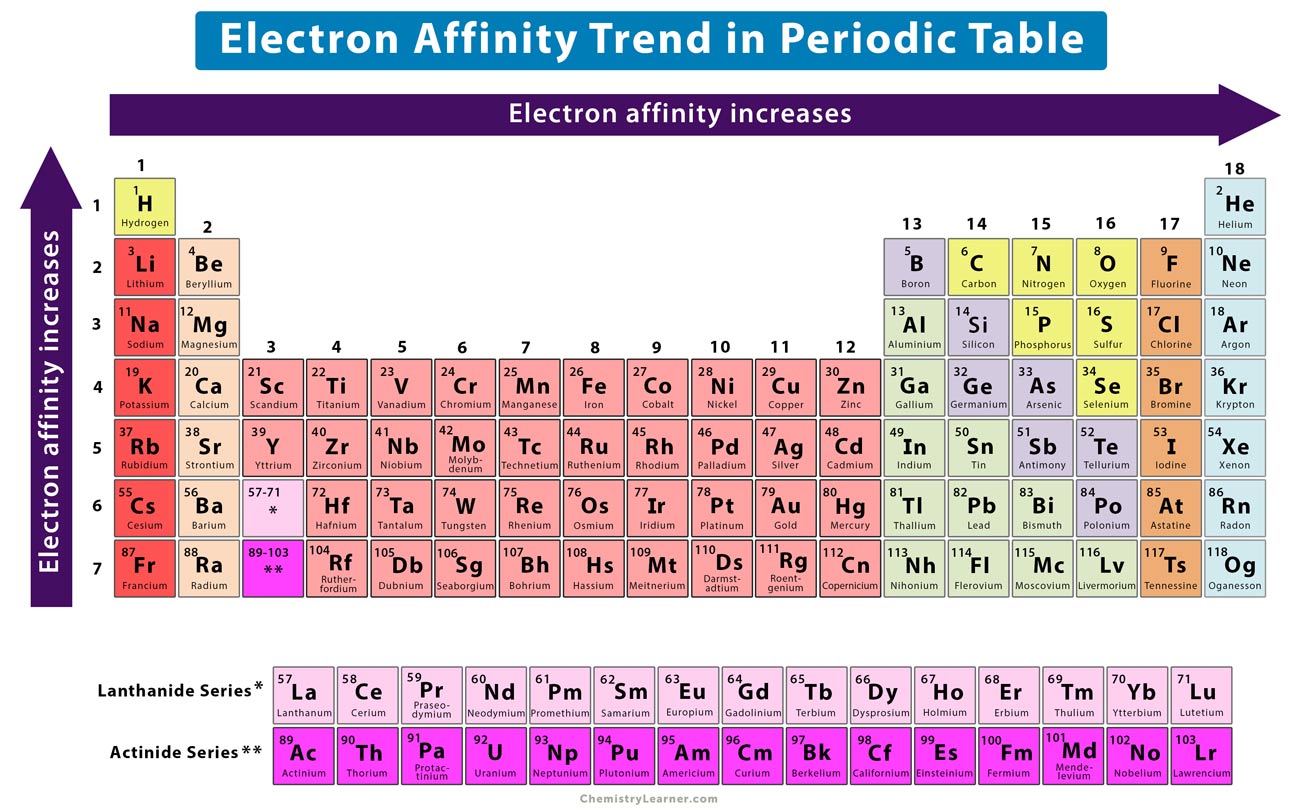
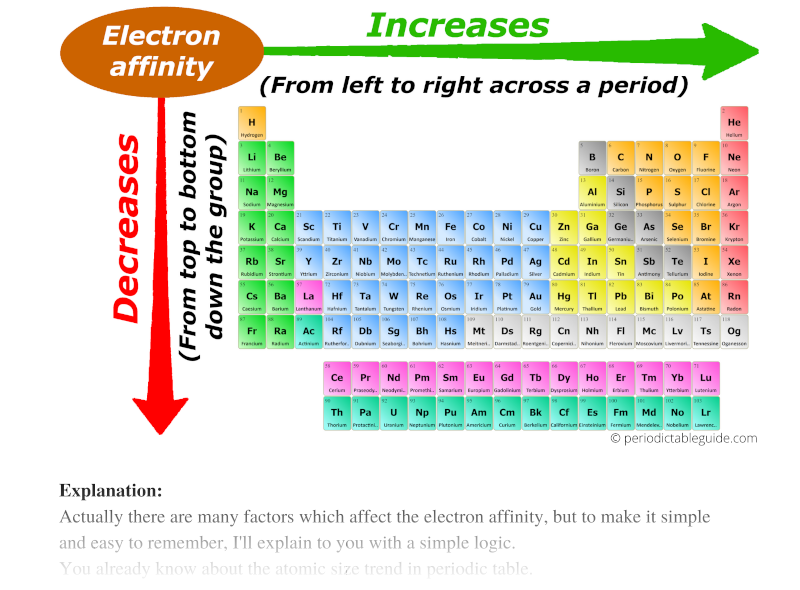
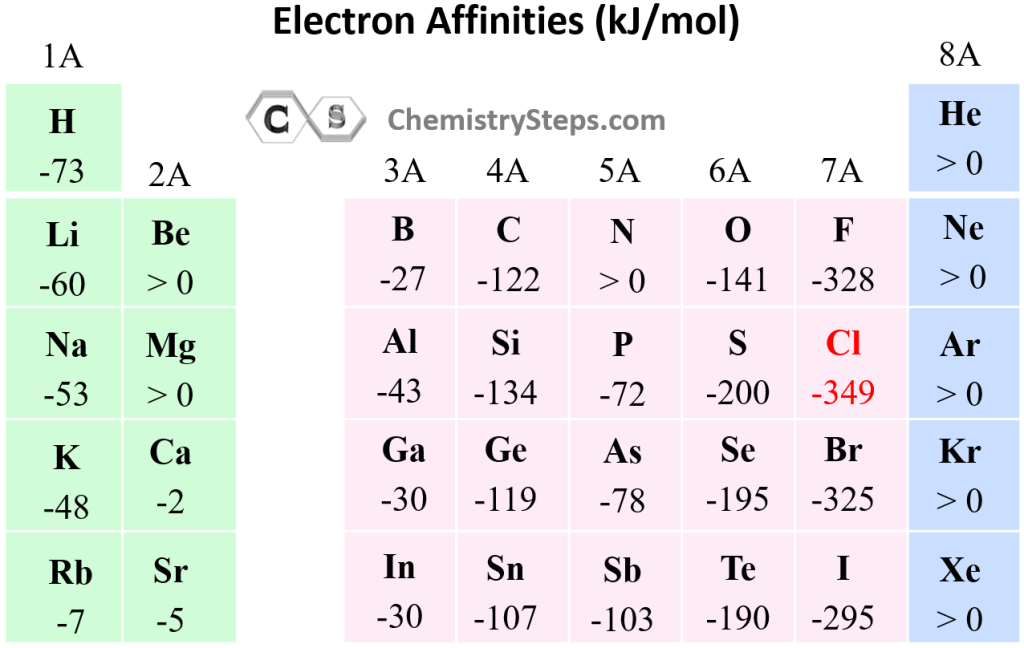
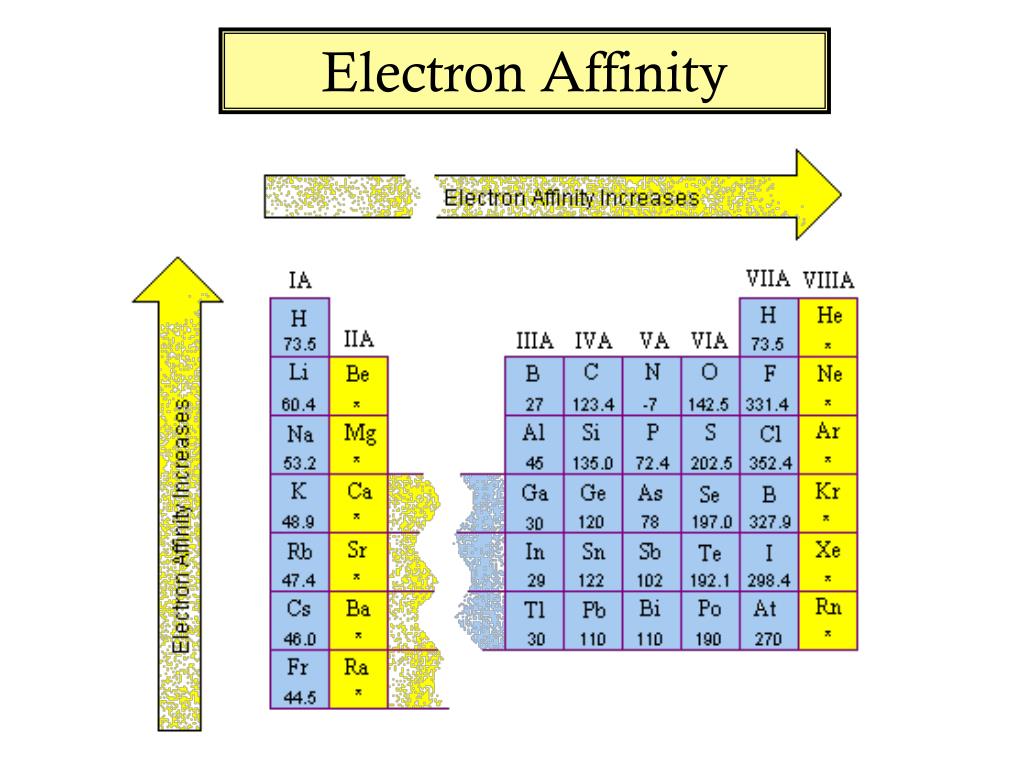
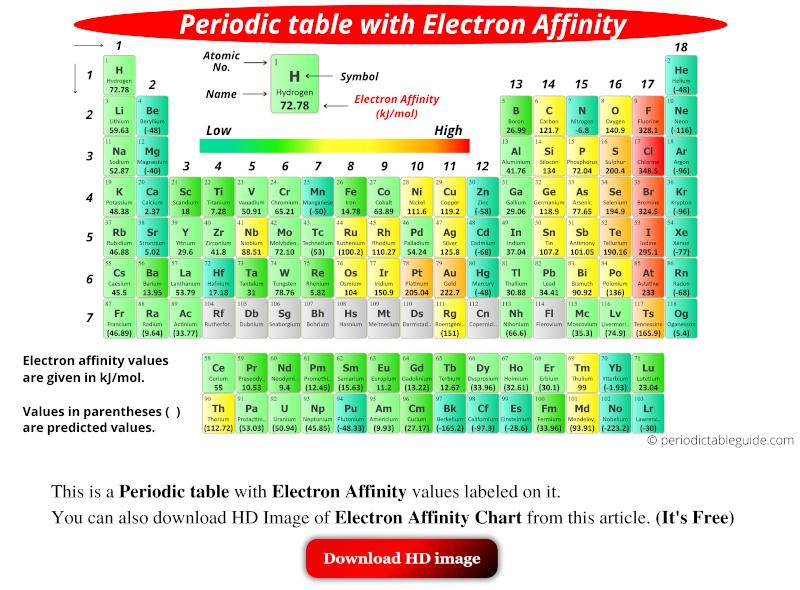
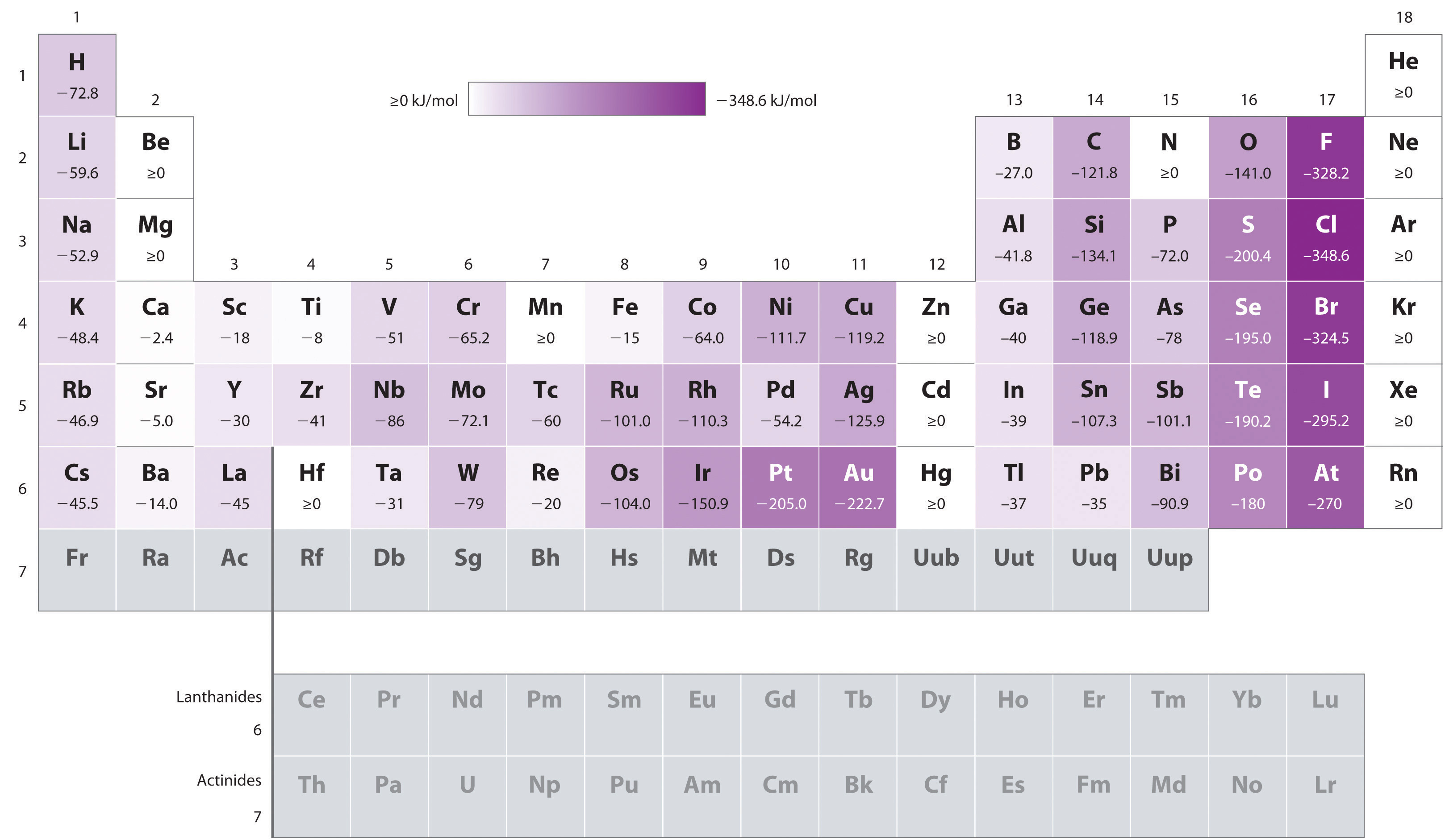
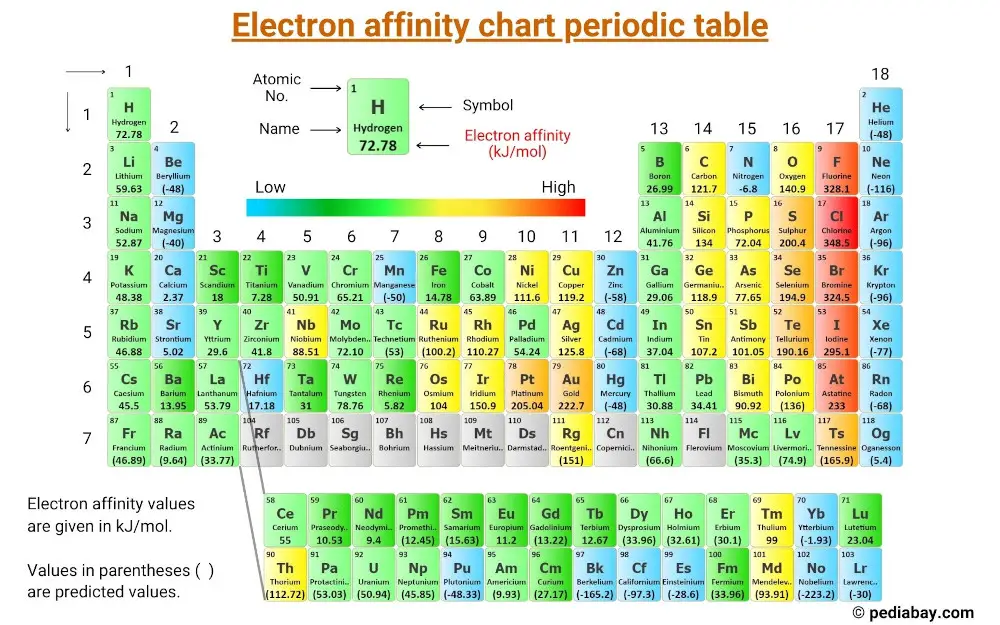
Chart Of Electron Affinity
Chart Of Electron Affinity - Groups via and viia in the periodic table have the largest electron affinities. First electron affinities have negative values. In general, elements with the most negative electron affinities (the highest affinity for an added electron) are those with the smallest size and highest ionization energies and are located in the upper right. The amount of energy released when an electron is added to atom) for most of chemical elements. Web the electron affinity is defined as the amount of energy released per mole when an electron is added to a neutral atom. Web the energy change that occurs when a neutral atom gains an electron is called its electron affinity. In general successive electron affinity increase in magnitude ea 1 < ea 2 < ea 3 and so on. How to find electron affinity Small numbers indicate that a less stable negative ion is formed. The second (reverse) definition is that electron affinity is the energy required to remove an electron from a singly charged gaseous negative ion. So the more negative the electron affinity the more favourable the electron addition process is. Electron affinity is the energy change when an atom gains an electron. The first electron affinities of the group 7 elements. Web the electron affinity is the potential energy change of the atom when an electron is added to a neutral gaseous atom to form a negative ion. For most elements, except noble gases, this is an exothermic process. Web the electron affinity (ea) of an element is the energy change that occurs when an electron is added to a gaseous atom to give an anion. Web table shows electron affinity (i.e. Web the most common units for electron affinity are kilojoules per mole (kj/mol) or electronvolts (ev). In general, elements with the most negative electron affinities (the highest affinity for an added electron) are those with the smallest size and highest ionization energies and are located in the upper right. The figure below shows electron affinities in \(\ce{kj/mol}\) for the representative elements. Web electron affinity chart for all the elements are given below. Web electron affinity is the energy change that results from adding an electron to a gaseous atom. By convention, the negative sign shows a release of energy. You can also get the periodic table labeled with electron affinity values of elements. In general successive electron affinity increase in magnitude. Web electron affinity is the energy change that results from adding an electron to a gaseous atom. In general, elements with the most negative electron affinities (the highest affinity for an added electron) are those with the smallest size and highest ionization energies and are located in the upper right. Web the most common units for electron affinity are kilojoules. The amount of energy released when an electron is added to atom) for most of chemical elements. In general successive electron affinity increase in magnitude ea 1 < ea 2 < ea 3 and so on. Web electron affinity is defined as the change in energy (in kj/mole) of a neutral atom (in the gaseous phase) when an electron is. The amount of energy released when an electron is added to atom) for most of chemical elements. Web explore how electron affinity changes with atomic number in the periodic table of elements via interactive plots. \ [x_ { (g)} + e^− \rightarrow x^−_ { (g)} + energy\] z. In general, elements with the most negative electron affinities (the highest affinity. Web the electron affinity is defined as the amount of energy released per mole when an electron is added to a neutral atom. In other words, the neutral atom's likelihood of gaining an electron. In general successive electron affinity increase in magnitude ea 1 < ea 2 < ea 3 and so on. When energy is released in a chemical. As discussed, electron affinities increase from left to right across periods; The amount of energy released when an electron is added to atom) for most of chemical elements. Web the energy change that occurs when a neutral atom gains an electron is called its electron affinity. \ [x_ { (g)} + e^− \rightarrow x^−_ { (g)} + energy\] z. In. The second (reverse) definition is that electron affinity is the energy required to remove an electron from a singly charged gaseous negative ion. Web electron affinity for all the elements in the periodic table. Web electron affinity is the energy change that results from adding an electron to a gaseous atom. In general, elements with the most negative electron affinities. Web electron affinity chart for all the elements are given below. The first electron affinities of the group 7 elements. First, as the energy that is released by adding an electron to an isolated gaseous atom. Web electron affinity can be defined in two equivalent ways. Web table shows electron affinity (i.e. These values are in kj/mol and the values written in parentheses ( ) are the predicted values. Web the electron affinity is defined as the amount of energy released per mole when an electron is added to a neutral atom. First, as the energy that is released by adding an electron to an isolated gaseous atom. Web table shows electron. The figure below shows electron affinities in \(\ce{kj/mol}\) for the representative elements. Electron affinity (ev) electron affinity (kj/mol) 1. In general successive electron affinity increase in magnitude ea 1 < ea 2 < ea 3 and so on. How to find electron affinity Groups via and viia in the periodic table have the largest electron affinities. Web electron affinity for all the elements in the periodic table. Electron affinity also applies to molecules, in some cases. In general successive electron affinity increase in magnitude ea 1 < ea 2 < ea 3 and so on. Web the electron affinity (e ea) of an atom or molecule is defined as the amount of energy release when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion. So the more negative the electron affinity the more favourable the electron addition process is. For most elements, except noble gases, this is an exothermic process. Web the most common units for electron affinity are kilojoules per mole (kj/mol) or electronvolts (ev). Web the electron affinity is defined as the amount of energy released per mole when an electron is added to a neutral atom. How to find electron affinity Web the electron affinity (ea) of an element is the energy change that occurs when an electron is added to a gaseous atom to give an anion. Web the electron affinity (ea) is the energy released to add an electron to an elements in the gaseous state. These values are in kj/mol and the values written in parentheses ( ) are the predicted values. Groups via and viia in the periodic table have the largest electron affinities. When energy is released in a chemical reaction or process, that energy is expressed as a negative number. Web electron affinity chart for all the elements of periodic table is shown in the below table. The second (reverse) definition is that electron affinity is the energy required to remove an electron from a singly charged gaseous negative ion.Periodic Behavior Presentation Chemistry
How about electron affinity? Electron affinity, Ionization energy
Electron Affinity Definition, Chart & Trend in Periodic Table
All Periodic Trends in Periodic Table (Explained with Image)
Electron Affinity Chemistry Steps
PPT Chapter 4 The Periodic Table PowerPoint Presentation, free
Electron Affinity Chart (Labeled Periodic table + List)
1.1.2.4 Electron Affinity Chemistry LibreTexts
Electron Affinity Chart of Elements (With Periodic Table) Pediabay
Electron Affinity Trend and Definition
Web The Electron Affinity Is The Potential Energy Change Of The Atom When An Electron Is Added To A Neutral Gaseous Atom To Form A Negative Ion.
Below Is A Visual Representation Of Electron Affinity Trends Throughout The Periodic Table.
Web The Electron Affinity (Ea) Of An Element Is The Energy Change That Occurs When An Electron Is Added To A Gaseous Atom To Give An Anion.
By Convention, The Negative Sign Shows A Release Of Energy.
Related Post:
.PNG)